Reproductive TechnologyReproduction is a natural method for animals to pass on genes to the next generation. Nowadays the reproductive process for mammals can be artificially carried out outside the body.
The main research interest in our lab is the development of reproductive technology in mammals. Meanwhile, we also work on deciphering the reproductive mechanism in relation to health and disease. The techniques that we have developed can be applied to enable the efficient production, preservation and distribution of genetically engineered mice in mouse repositories. We wish to share the knowledge that we have accumulated through our research into mouse reproductive biology with the scientific community, so all of our protocols are published on our website. We offer support to researchers using our techniques to conduct experiments using genetically engineered mice. |
|
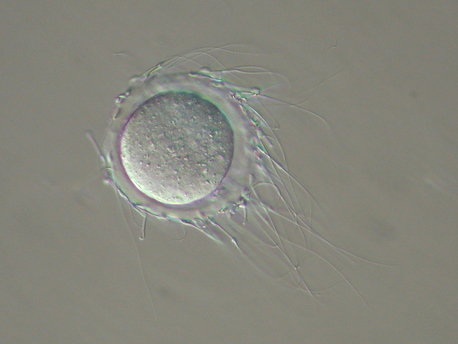
In vitro fertilization
|
Pharmaceutically assisted IVF
First successful example of in vitro fertilization (IVF) in mice was reported in 1968. The conditions for IVF have been optimized since then in order to stabilize and enhance fertilization rate. In 2008, we reported that methyl-beta-cyclodextrin (MBCD) had great ability to improve the fertilizing ability of cryopreserved C57BL/6 mice sperm (Takeo et al, Biol Reprod, 2008). Furthermore, in 2011 we reported that reduced glutathione (GSH) promoted fertilization by facilitating sperm penetration through the zona pellucida of mouse oocytes (Takeo and Nakagata, Biol Reprod, 2011). In addition, we demonstrated cysteine derivatives also enhanced the fertilizing ability of oocytes (Takeo et al, Biol Reprod, 2015). Now, the compounds can be found in mouse sperm preincubation and fertilization medium. |
|
Cryotechnology
|
Embryo cryopreservation
Embryo cryopreservation has been traditionally used to efficiently maintain and preserve mouse lines in mouse banks. There are several benefits to the preservation of mouse lines via embryo cryopreservation. For example, using the technology we can obtain offspring that carry the same genotypes; drastically reduce the risk of disaster, fire, and genetic contamination; save on the space, labor, and breeding cost; and ship mice to other researchers more simply. We have developed a cryopreservation method via vitrification using DAP213 (2M DMSO, 1M Acetamide and 3M propylene glycol, Nakao et al, Exp Animal, 1997). The vitrification method also brings many benefits: the procedure is very simple and quick; we do not need any special devices to control the cooling rate; and we can obtain a high and stable rate of survival. Sperm cryopreservation Sperm cryopreservation is the most efficient means of archiving genetically engineered mice in a mouse resource bank. The benefits of sperm cryopreservation include the efficient storage and shipment of mice lines, an easier procedure than for embryo cryopreservation, quick preservation without special devices or skills, and the ability to produce more than 2,000 mice from straws derived from a single male. We have established a robust protocol of mouse sperm cryopreservation using 18% raffinose pentahydrate and 3% skim milk (R18S3). This is called the Nakagata Method (Nakagata, Mammal Genome, 2000). Nowadays, we use a modified protocol using mR18S3 which contains L-glutamine in R18S3 (Takeo and Nakagata, Lab Anim, 2010). Oocyte cryopreservation Oocyte cryopreservation provides an efficient method of preserving genetic resources derived from female mice, and also offers a useful source of oocyte donors to produce the intended progeny of genetically engineered mice via IVF. The benefits of oocyte cryopreservation include the efficient storage and shipment of mice lines; minimal rearing periods for oocyte donors; and the ability to use oocytes at any time. We have published several papers regarding mouse oocyte cryopreservation via the vitrification method (Nakagata, J Reprod Fertil, 1989) and IVF between the oocytes and fresh, cryopreserved or cold-stored sperm (Nakagata et al, Cryobiology, 2013). Cold storage of embryos The cold storage of 2-cell mouse embryos offers an alternative method of transporting genetically engineered mice to the transport of live animals or of cryopreserved embryos or sperm. The advantages of using cold-stored embryos are that it eliminates the need to learn special techniques for handling samples; the embryos can be transported under refrigerated temperatures; and the cost of shipment is cheaper than that of live animals or frozen samples. In addition, the shipment of cold-stored embryos is newly applicable to disseminate archived embryos of genetically engineered mice from mouse resource banks. We have developed a cold transport system for 2-cell mouse embryos (Takeo et al, Cryobiology, 2009). We found that the M2 medium maintained the viability and developmental ability of 2-cell embryos well at refrigerated temperatures for up to 72 hours. Recently, we prolonged the storage period of 2-cell embryos by storing in M2 medium with N-acetyl-cysteine (Horikoshi et al, Cryobiology, 2016). Cold storage of sperm The cold storage of mouse sperm is the simplest means of transporting genetically engineered mice. The advantages of this method are that it is easy to collect samples; it allows for the efficient shipment of samples; the sperm remains viable for longer than embryos at refrigerated temperatures; and cold-stored sperm can be cryopreserved after arrival. We have developed a cold transport system for sperm enclosed in a cauda epididymis (Takeo et al, Cryobiology, 2012). The sperm transported at refrigerated temperatures can be cryopreserved and used in IVF after thawing (Takeo et al, Cryobiology, 2014). Methyl-beta-cyclodextrin improved the fertility of cold-stored sperm by inducing acrosome reaction and hyperactivation with membrane destabilization after cholesterol efflux (Yoshimoto et al, Biol Reprod, 2017). |
|
Superovulation
|
Ultra superovulation
The number of ovulated oocytes is regulated by hypothalamic-pituitary-ovarian axis interaction with hormones. Superovulation is carried out via the administration of hormones, which results in the promotion of follicular development and ovulation. In general, equine chorionic gonadotropin (eCG) is administered to stimulate follicular development, and then human chorionic gonadotropin (hCG) is injected to induce superovulation in mice. The technique using eCG and hCG can generally generate 20 oocytes per female mouse. However, in 2015 we reported that more than 100 oocytes were generated via the combined administration of inhibin antiserum (IAS) and eCG (Takeo and Nakagata, PLoS One, 2015). This novel superovulation technique generated 5 times more oocytes than the conventional method using eCG and hCG. The ultrasuperovulation technique was effective among various inbred and outbred mice (Takeo and Nakagata, Theriogenology, 2016). |
|
Genome Editing
|
CRISPR/CAS and TALEN
Genome editing techniques using Clustered regulatory interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9) or Transcription activator-like effector nuclease (TALEN) allows the rapid and efficient production of genetically engineered mice. To generate the mice, fertilized oocytes were used to manipulate target gene by the techniques. We published an efficient procedure of knockout mice by CRISPR/CAS system using cryopreserved oocytes at pronuculeus stage (Nakagawa et al, BMC Biotechnology, 2015). Recently, we produced various mutant mice derived from ultrasuperovulation technique and CRISPR/CAS system (Nakagawa et al, Biology Open, 2016). |
Mouse Resource |
Reproductive Technology is a powerful tool to efficiently manage genetic resource. Since 1998, we provide a service of CARD Mouse Bank using our developed techniques of mouse reproductive technology at Center for Animal Resources and Development (CARD), Kumamoto University. Aim of CARD Mouse Bank is to accelerate research on medical and life science by supporting production, preservation, and supply of useful mouse models.
|
Animal Welfare |
Improvement of reproductive technology is directly contributed to reduce the number of laboratory animals. We achieved to reduce the number of oocyte donors by improving the techniques of superovulation using inhibin antiserum and gonadotropins and in vitro fertilization using methyl-beta-cyclodextrin and reduced glutathione.
|